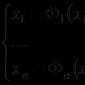Let's imagine the following situation:
You work in a lab and decide to do an experiment. To do this, you opened the cabinet with reagents and suddenly saw the following picture on one of the shelves. Two jars of reagents had their labels peeled off, which were safely left lying nearby. At the same time, it is no longer possible to determine exactly which jar corresponds to which label, and the external signs of the substances by which they could be distinguished are the same.
In this case, the problem can be solved using the so-called qualitative reactions.
Qualitative reactions called such reactions that allow you to distinguish one substance from another, as well as to find out the qualitative composition of unknown substances.
For example, it is known that the cations of some metals, when their salts are added to the burner flame, color it in a certain color:
This method can work only if the substances to be distinguished change the color of the flame in different ways, or one of them does not change color at all.
But, let's say, as luck would have it, the substances you determine do not color the color of the flame, or they color it in the same color.
In these cases, it will be necessary to distinguish substances using other reagents.
In what case can we distinguish one substance from another with the help of any reagent?
There are two options:
- One substance reacts with the added reagent, while the other does not. At the same time, it must be clearly seen that the reaction of one of the starting substances with the added reagent has really passed, that is, some external sign of it is observed - a precipitate precipitated, gas was released, a color change occurred, etc.
For example, it is impossible to distinguish water from a sodium hydroxide solution using hydrochloric acid, despite the fact that alkalis react perfectly with acids:
NaOH + HCl \u003d NaCl + H 2 O
This is due to the absence of any external signs of a reaction. A transparent colorless solution of hydrochloric acid, when mixed with a colorless hydroxide solution, forms the same transparent solution:
But on the other hand, water can be distinguished from an aqueous solution of alkali, for example, using a solution of magnesium chloride - a white precipitate forms in this reaction:
2NaOH + MgCl 2 = Mg(OH) 2 ↓+ 2NaCl
2) substances can also be distinguished from each other if they both react with the added reagent, but do so in different ways.
For example, a solution of sodium carbonate can be distinguished from a solution of silver nitrate using a solution of hydrochloric acid.
Hydrochloric acid reacts with sodium carbonate to give off a colorless, odorless gas. carbon dioxide(CO 2):
2HCl + Na 2 CO 3 \u003d 2NaCl + H 2 O + CO 2
and with silver nitrate to form a white cheesy precipitate AgCl
HCl + AgNO 3 \u003d HNO 3 + AgCl ↓
The tables below show different options for detecting specific ions:
Qualitative reactions to cations
| Cation | Reagent | Sign of reaction |
| Ba 2+ | SO 4 2- |
Ba 2+ + SO 4 2- \u003d BaSO 4 ↓ |
| Cu2+ | 1) Precipitation of blue color: Cu 2+ + 2OH - \u003d Cu (OH) 2 ↓ 2) Precipitation of black color: Cu 2+ + S 2- \u003d CuS ↓ |
|
| Pb 2+ | S2- | Precipitation of black color: Pb 2+ + S 2- = PbS↓ |
| Ag+ | Cl- |
Precipitation of a white precipitate, insoluble in HNO 3, but soluble in ammonia NH 3 H 2 O: Ag + + Cl − → AgCl↓ |
| Fe2+ |
2) Potassium hexacyanoferrate (III) (red blood salt) K 3 |
1) Precipitation of a white precipitate that turns green in air: Fe 2+ + 2OH - \u003d Fe (OH) 2 ↓ 2) Precipitation of a blue precipitate (turnbull blue): K + + Fe 2+ + 3- = KFe↓ |
| Fe3+ |
2) Potassium hexacyanoferrate (II) (yellow blood salt) K 4 3) Rhodanide ion SCN − |
1) Precipitation of brown color: Fe 3+ + 3OH - \u003d Fe (OH) 3 ↓ 2) Precipitation of a blue precipitate (Prussian blue): K + + Fe 3+ + 4- = KFe↓ 3) The appearance of intense red (blood red) staining: Fe 3+ + 3SCN - = Fe(SCN) 3 |
| Al 3+ | Alkali (hydroxide amphoteric properties) |
Precipitation of a white precipitate of aluminum hydroxide when a small amount of alkali is added: OH - + Al 3+ \u003d Al (OH) 3 and its dissolution upon further addition: Al(OH) 3 + NaOH = Na |
| NH4+ | OH − , heating | Emission of gas with a pungent odor: NH 4 + + OH - \u003d NH 3 + H 2 O Blue wet litmus paper |
| H+ (acid environment) |
Indicators: − litmus − methyl orange |
Red staining |
Qualitative reactions to anions
| Anion | Impact or reagent | Reaction sign. Reaction equation |
| SO 4 2- | Ba 2+ |
Precipitation of a white precipitate, insoluble in acids: Ba 2+ + SO 4 2- \u003d BaSO 4 ↓ |
| NO 3 - |
1) Add H 2 SO 4 (conc.) and Cu, heat 2) A mixture of H 2 SO 4 + FeSO 4 |
1) Formation of a blue solution containing Cu 2+ ions, brown gas evolution (NO 2) 2) The appearance of the color of nitroso-iron sulfate (II) 2+. Violet to brown color (brown ring reaction) |
| PO 4 3- | Ag+ |
Precipitation of a light yellow precipitate in a neutral medium: 3Ag + + PO 4 3- = Ag 3 PO 4 ↓ |
| CrO 4 2- | Ba 2+ |
Precipitation of a yellow precipitate, insoluble in acetic acid, but soluble in HCl: Ba 2+ + CrO 4 2- = BaCrO 4 ↓ |
| S2- | Pb 2+ |
Black precipitation: Pb 2+ + S 2- = PbS↓ |
| CO 3 2- |
1) Precipitation of a white precipitate, soluble in acids: Ca 2+ + CO 3 2- \u003d CaCO 3 ↓ 2) Emission of a colorless gas ("boiling"), causing the lime water to become cloudy: CO 3 2- + 2H + = CO 2 + H 2 O |
|
| CO2 | Lime water Ca(OH) 2 |
Precipitation of a white precipitate and its dissolution upon further passage of CO 2: Ca(OH) 2 + CO 2 = CaCO 3 ↓ + H 2 O CaCO 3 + CO 2 + H 2 O \u003d Ca (HCO 3) 2 |
| SO 3 2- | H+ |
SO 2 gas evolution with a characteristic pungent odor (SO 2): 2H + + SO 3 2- \u003d H 2 O + SO 2 |
| F- | Ca2+ |
Precipitation of a white precipitate: Ca 2+ + 2F - = CaF 2 ↓ |
| Cl- | Ag+ |
Precipitation of a white cheesy precipitate, insoluble in HNO 3 but soluble in NH 3 H 2 O (conc.): Ag + + Cl - = AgCl↓ AgCl + 2(NH 3 H 2 O) =)
Most Discussed
 |